|
4/10/2024 0 Comments Water density lbgalWhat is the weight in lbs/cf of a substance that has a specific gravity of 2.05?Ī shipment of crude oil has a specific gravity of 0.825. How many pounds of ferric chloride are in 250 gallons of 22% strength? (Assume the specific gravity is 1.41) Chemical A is what percent heavier than chemical B? What is the specific gravity?Ĭhemical A has a specific gravity of 2.31 and chemical B has a specific gravity of 1.95. density of water, distilled is equal to 1 000 kg/m³ at 25☌ (77☏ or 298.15K) at. 1 cubic meter of Water, distilled weighs 1 000 kilograms kg 1 cubic foot of Water, distilled weighs 62.42796 pounds lbs Water, distilled weighs 1 gram per cubic centimeter or 1 000 kilogram per cubic meter, i.e. How much does 45 gallons of 25% Alum weigh if the specific gravity is 1.35?įerric chloride weighs 14.25 lbs/gal. There are 8.35 pounds in a gallon of water. How many pounds of the 50 gallons are available chlorine? The equation is based on water with temperature 68oF (20oC) and specific weight 8.33 lb/US gal. What is the weight difference between 75 gallons of water and 42 gallons of sodium hypochlorite with a specific gravity of 1.42?Ī treatment operator has 50 gallons of 12.5% sodium hypochlorite. Table 14 Calcium Chloride Brine Table (Properties at 20° C) WTc. What is the corresponding weight in pounds per gallon?Ĭhlorine gas has formed into a liquid state. Liquid sodium hypochlorite has a specific gravity of 1.47. Solve the following density related problems. On any State exam, you will be given the specific gravity or corresponding weight of the substance in the question. Remember, these are only examples and should not be put to memory. The table below lists common specific gravities/densities and weight of substances used in the waterworks industry. Conversely, if a substance has a specific gravity greater than 1, it will sink in water. For example, if something has a specific gravity less than water (<1), then the substance will float on water. When discussing specific gravity, many things are compared to water. This is also referred to as specific gravity. 1 pound per gallon US ( lb/gal ) 7.48 pounds per cubic foot ( lb/ft3 ). The density (mass per unit volume) of water is 1.00. This on the web one-way conversion tool converts density units from pounds per gallon US ( lb/gal ) into pounds per cubic foot ( lb/ft3 ) instantly online. Substituting the density of water and the volume, we get: Mass pounds 8.3454 pounds/gallon × 5 gallons 41.73 pounds. 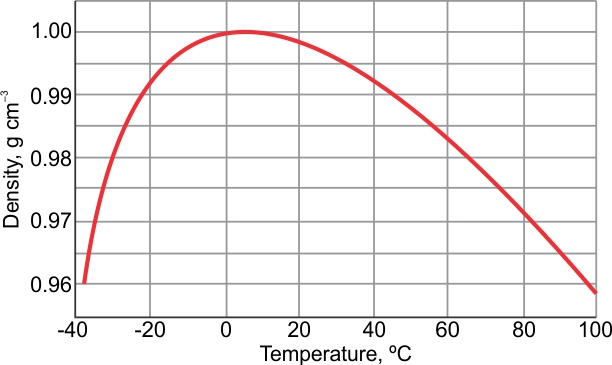
So, how much does water actually weigh? There are a few variables, such as temperature, that determine the weight of water, but for all practical purposes in waterworks mathematics, water weighs 8.34 pounds per gallon. The answer to how many pounds are in 5 gallons of water comes from the widely known mass-density-volume formula: Mass pounds Density pounds/gallon × Volume gal. The same density of water is 1.0249 g/ml or 63.982 lb/ft³. And thats it The density of salt water is 1,024.9 kg/m³. and kilogram/litre.You can view more details on each measurement unit: pounds/gallon or kg/l The SI derived unit for density is the kilogram/cubic meter. We assume you are converting between pound/gallon U.S. 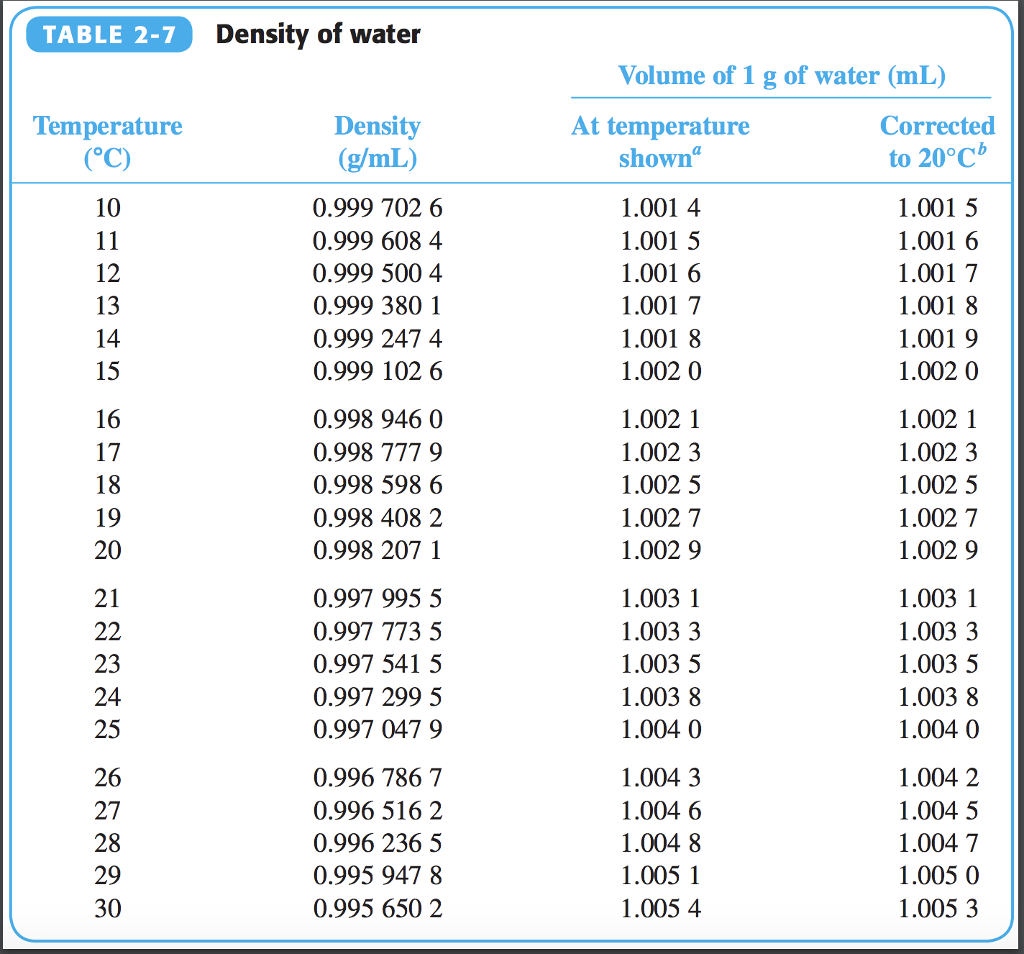
How many pounds/gallon in 1 kg/l The answer is 8.3454042651525. 
Example: Class A cement plus 4 bentonite using normal mixing water: Determine the following: Amount of bentonite to add Total water requirements. More information from the unit converter. It makes up approximately 60% of our total body weight and as much as 75% of the earth’s surface. What is the density of water at 20 ☌ of salinity S 35 and under the pressure of 1 atm Lets give our water density calculator a try and find out Set the temperature to 20 ☌. e) Slurry density, lb/gal: Density, lb/gal 94 + wt of additive + (8.33 x vol. Water is essential for the survival of all life forms. Pruß: The IAPWS Formulation 1995 for the Thermodynamic Properties of Ordinary Water Substance for General and Scientific Use. * - NIST Standard Reference Database 69: NIST Chemistry WebBook, Thermophysical Properties of Fluid Systems, Isobaric Properties for Water, 2008. * Temperature according to ITS-90 (International Temperature Scale from 1990). The specific gravity of PG, the density of which is 1.037 at 77☏ (25☌), when compared to water, which has density of 0. The following table lists the density D of water as a function of the temperature under isobaric conditions : Densities, and therefore specific gravities, vary with temperature hence it is necessary to indicate the temperatures of both the glycol and water.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
